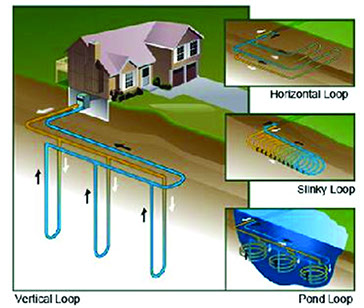
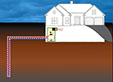
How a geothermal heat exchanger uses the refrigeration process to heat your home.
The second law of thermodynamics states that the entropy of an isolated system never decreases, because isolated systems spontaneously evolve toward thermodynamic equilibrium—the state of maximum entropy. Wikipedia
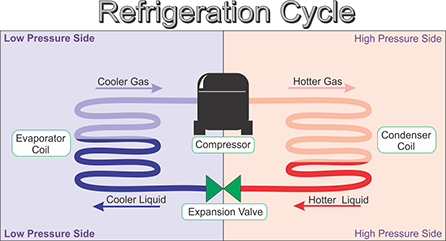
The refrigeration process is simply a cycle where a substance is picks up (absorbs) heat energy from one part of the cycle and gets rid of that heat energy in another part.
What the heck is entropy?
And why do I care??
There is no quiz, so you do not need to know everything about the refrigeration process. Just a few things, though, will help you to understand how a geothermal heat pump works.
Just follow this explanation to the bottom.
1) Heat energy is transferred by one of three ways:
Conduction (Through a solid or a liquid)
Convection (Through air transfer)
Radiation (Infrared: Sun, wood stove, radiant floor heat)

2) Heat energy will flow from a hotter object to a colder object spontaneously. All by itself. No effort needed.

3) Compressing a gas makes it hotter.
This is an example of DECREASING entropy. You always need to expend energy to make this happen. When you squeeze, compress a gas into a smaller space, that gas gets hotter.

Note the cooling fins on any air compressor.
4) Expanding a gas makes it colder.
This is an example of DECREASING entropy. Basically, this will happen all by itself. Gas will expand unless confined. The molecules of gas get further apart and that makes the gas colder. Things will cool off to their surroundings temperature unless a heat source is applied.

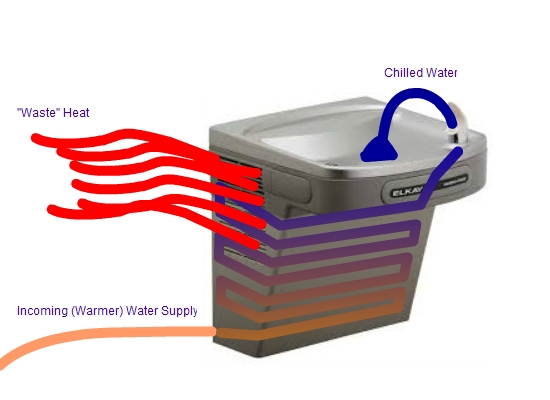
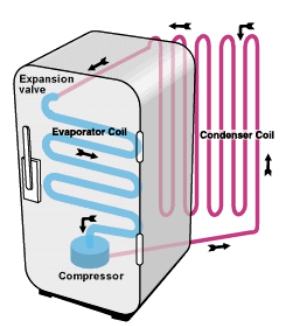





5) Through the refrigeration cycle, the colder side absorbs heat from it's warmer surroundings and the hotter side rejects heat to it's cooler surroundings.
These are all doing the same thing. Moving heat energy from one place to the other. Take special note of the water cooler (bubbler, to those in the southeast part of the state).
As the drinking water moves through the fountain, it becomes chilled.
How?
The refrigeration process in this case absorbs the heat energy from the water (through conduction) because the tube is immersed in an ice bath and rejects it (through convection by blowing warm air) into the hallway.

When you put a warm bottle of beer in an ice bath, that beer becomes colder. The heat energy from the liquid is transferred into the ice, melting it.

Think of it this way:
Cold water returning to the ground loop.
Warm air circulating through your house.
Warmer water coming from the ground loop.
6) Conduction is a much more efficient method of heat transfer than convection.
I think that it is safe to say that this man will lose a great deal more heat energy from his body after he is in contact (Conduction) with the water than while in the air (Convection).

Meaning THIS:
Transfers heat much more efficiently than THIS:

Or THIS:

So that is essentially how a geothermal heat pump uses the refrigeration process to easily move heat energy from a warmer place (the Earth in the winter) to your home and from you hot home to a relatively cool place (the Earth in the summer).
Because a geo system does not generate heat, it only moves heat, it is very, very efficient, from an energy standpoint. How can you say that it is more than 100% efficient? That sounds crazy!
In a gas furnace, for every one unit of energy consumed, you get a large part of that returned as heat.


In the above example, 96% is returned as heat, which is great!
Electric resistance heat is virtually 100% efficient. That means for every one unit of energy consumed, you get one unit of heat energy returned.
A geothermal heat pump is 300% or more efficient. That means for every one unit of energy consumed, you get three or four or five or more units of heat energy returned.

Bonus Chart: Why phase change stores and releases so much energy!
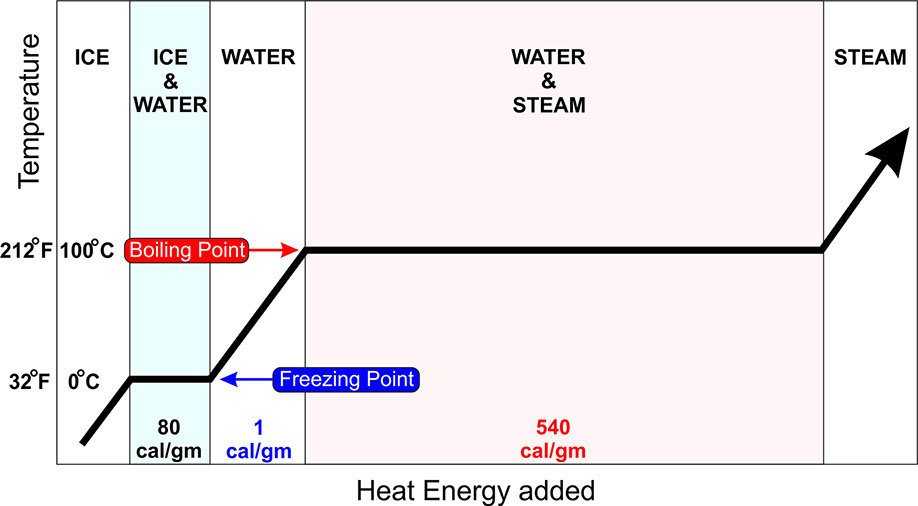
You add 1 calorie to raise 1 gram of water 1 degree.
You add 540 calories to raise 1 gram of water ZERO degrees. Just to change from a liquid to a gas. That same energy is released when that gas condenses to a liquid.

